-
PDF
- Split View
-
Views
-
Cite
Cite
NENA DE G. ALVAREZ, ROYDON J. MEEKING, DEREK W. R. WHITE, The Origin, Initiation and Development of Axillary Shoot Meristems in Lotus japonicus, Annals of Botany, Volume 98, Issue 5, November 2006, Pages 953–963, https://doi.org/10.1093/aob/mcl187
Close - Share Icon Share
Abstract
• Background and Aims Lotus japonicus ‘Gifu’ develops multiple axillary shoots in the cotyledonary node region throughout the growth of the plant. The origin, initiation and development of these axillary meristems were investigated.
• Methods Morphological, histological and mRNA in situ analyses were done to characterize the ontogeny of cotyledonary axillary shoot meristems in Lotus. Morphological characterization of a putative Lotus shoot branching mutant (super-accessory branches) sac, is presented.
• Key Results By using expression of an L. japonicus STM-like gene as a marker for meristematic tissues, it was demonstrated that groups of cells maintained in the meristematic state at the cotyledonary axil region coincide with the sites where additional axillary meristems (accessory meristems) form. A Lotus shoot branching mutant, sac, is a putative Lotus branching mutant characterized by increased proliferation of accessory shoots in all leaf axils including the cotyledons.
• Conclusion. In Lotus, axillary shoot meristems continually develop at the cotyledonary node region throughout the growth of the plant. These cotyledonary primary and accessory axillaries arise from the position of a meristematic zone of tissue at the cotyledonary node axil region.
INTRODUCTION
Variation in branching pattern is one of the main factors contributing to the vast diversity in shoot architecture found amongst plants. This variability is in turn influenced by patterns of axillary shoot meristem initiation, lateral shoot bud development and lateral shoot branch outgrowth. Among the flowering plants, shoot branches are derived from axillary meristems initiated in the axils of leaves (Steeves and Sussex, 1989). Axillary meristems, which subsequently function as shoot apical meristems (SAMs), provide plants with unlimited growth potential. As long as the plant maintains a group of these meristematic cells in the vegetative state, shoot growth and development is indeterminate and the plant has the potential for an open-ended perennial lifespan.
The model plants Arabidopsis thaliana and Lotus japonicus have different patterns of axillary meristem initiation and development. In Arabidopsis, axillary buds are not morphologically evident in the axils of rosette leaves during the vegetative stage of plant development (Hempel and Feldman, 1994), unless the plants grow vegetatively for a prolonged period, in which case development of axillary meristems is activated first at the basal-most nodes and progresses in an acropetal fashion (Grbic and Bleecker, 2000). Once the plant transitions from vegetative growth to flowering, axillary buds form and grow out in a basipetal wave from newly formed nodes near the shoot apex to the older basal nodes. These axillary meristems undergo a short vegetative phase before they undergo transition into reproductive development. Typically, a single axillary meristem initiates in a leaf axil, although the leaf axils may have the potential to produce multiple buds.
A different pattern of shoot branching is observed in L. japonicus. Axillary buds are morphologically visible at the cotyledonary node early during vegetative development of the plant. These cotyledonary axillary shoot buds develop immediately into lateral shoots in a pattern similar to development of the primary shoot. Throughout vegetative and reproductive development, additional axillary shoot meristems (accessory meristems) continue to be initiated in the cotyledonary node region, resulting in the development of numerous basal shoot branches.
Shoot branching has been extensively examined (Stafstrom et al., 1998; Schmitz and Theres, 1999; Grbic, 2001; Shimizu-Sato and Mori, 2001; Ward and Leyser, 2004; McSteen and Leyser, 2005; Schmitz and Theres, 2005; Bennet and Leyser, 2006), but the basic mechanisms regulating shoot branching remain unclear. Many of these studies on the regulation of shoot branching have centred on apical dominance and the role of endogenous plant hormones, particularly auxins (Cline, 1991, 1997; Li et al., 1992; Romano et al., 1993; Beveridge et al., 2000; Chatfield et al., 2000; Morris et al., 2005). Recent results suggests that auxins and cytokinins are involved not only in the outgrowth of buds but also in the specification of axillary meristems (Tantikanjana et al., 2001; Greb et al., 2003).
The characterization of mutants with defects in shoot branching has been a useful means of studying the regulation of axillary shoot development in plants (Napoli et al., 1999; Schmitz and Theres, 1999; Beveridge et al., 2003). The spectrum of branching mutants examined includes different plant species with defects in the initiation of axillary meristems, such as in Arabidopsis (McConnell, 1995; Otsuga, 2001; Greb et al., 2003), rice (Li et al., 2003), tomato (Malayer and Guard, 1964; Schmitz et al., 2002) and maize (Gallavotti et al., 2004), or in the outgrowth of lateral shoot buds (Stirnberg et al., 1999, 2002; Rameau et al., 2002; Sorefan et al., 2003; Takeda, 2003; Bainbridge et al., 2005; Snowden, 2005). The supershoot mutant of Arabidopsis belongs to yet another class of these mutants, and is characterized by a massive proliferation in the number of meristems formed in the leaf axil, together with release of bud arrest (Tantikanjana et al., 2001).
The discovery of genes such as SHOOT MERISTEMLESS (STM), whose transcript accumulation marks cells with a meristematic fate (Long et al., 1996), provides an effective tool for studying the early ontogeny of axillary meristems. STM, a member of a family of KN 1-like genes, is expressed throughout the SAM of Arabidopsis, but is downregulated in positions where leaves are initiated.
In this report, a description is given of the shoot branching pattern of L. japonicus. By morphological and anatomical analyses, information is provided about the origin and initiation of axillary meristems and their development into shoot branches. A putative Lotus homologue of Arabidopsis STM has also been isolated and the expression pattern of L. japonicus STM-like (LjSTM-like) gene transcripts has been followed to study the early ontogeny of axillary meristems in the cotyledonary node region of Lotus. Additionally, a preliminary morphological analysis of axillary shoot branching in a Lotus branching mutant, sac is presented in order to provide a framework within which the effects of branching genes could be studied in the future.
MATERIALS AND METHODS
Definition of accessory buds
Lotus has an unusual branching pattern where vegetative lateral shoot branches develop continually at both the cotyledonary node and aerial leaf axils throughout the development of the plant. The following definitions have been used to describe the type of axillaries formed in Lotus. A lateral bud generally refers to a macroscopically visible bud present at nodes. It may refer to a vegetative or inflorescence bud, an axillary or accessory bud. In this study, axillary bud is used to refer to the primary axillary bud (vegetative or inflorescence) growing in all leaf axils including that of the cotyledons. Multiple buds growing out alongside the primary axillary bud in the same leaf axil are referred to as accessory buds. Axillary buds growing out of the cotyledonary region are referred to as cotyledonary or basal axillary buds, while all axillary buds growing out of the nodes above the cotyledons are referred to as aerial axillary buds.
Plant material and growth conditions
Lotus japonicus S3343 (‘Gifu’) was used in this study. Seeds were first soaked in water overnight and sown directly in soil potting medium. The plants were grown in a controlled environment growth cabinet with a 14 h/10 h day/night cycle and a constant 20–21 °C day/night temperature. Growth measurements were made at weekly intervals. Seedlings were removed from the growth cabinet to record the appearance of lateral buds using ten representative plants. An axillary bud was considered to be present when the bud was morphologically visible under a dissecting microscope.
Plant tissue culture
The seedlings used for histological and scanning electron microscopy (SEM) analyses were grown in Murashige and Skoog (MS) medium under controlled environment conditions (14 h/10 h day/night cycle at an average room temperature of 24 °C). A week after sowing the seeds in culture, cotyledons were detached from some of the seedlings, laid flat on the culture medium and grown for a period of 2 months under the same conditions.
Histological analysis
Tissue samples were fixed with FAA for 3 h at room temperature. Fixed materials were dehydrated in a graded butanol series and embedded in paraffin wax (Paraplast Plus). Wax blocks were stored at 4 °C until further processing. Sections 8 µm thick were cut on a microtome, affixed to microscope slides, dewaxed in Histoclear, rehydrated through ethanol series and stained with Saffranin-Fast Green. These were mounted with a xylene-based mountant DPX after dehydration through ethanol and Histoclear. Microscope slides were examined under bright field with an Olympus BX50 light microscope and images were recorded using Fujichrome or a digital camera attached to an Olympus analySISB imaging system. Some of the images were captured with Nomarski optics.
Scanning electron microscopy
Plant materials were fixed in 4 % glutaraldehyde in 50 mm potassium phosphate buffer at pH 7·0, dehydrated through a graded ethanol series, critical point dried in CO2, sputter coated with gold and viewed under a scanning electron microscope.
Isolation and sequence analysis of a LjSTM-like gene
LjSTM-like cDNA was amplified by reverse transcription–polymerase chain reaction (RT–PCR) from total RNA isolated from shoot tips and cotyledonary nodes (the cotyledons were removed) of 7-day-old seedlings. The PCR STM primers used were designed using the sequence of a full-length cDNA Medicago truncatula knotted-like sequence recorded in the GenBank database (Author Webpage). PCR products were cloned in pGEM T or pGEM T 4Easy (Promega) plasmid vectors and inserts were sequenced using an automated sequencer (AB 310). Sequence alignment was done using Clustal W (Vector NTI program, Invitrogen).
In situ hybridization
Fresh plant materials were fixed with FAA (vacuum-infiltrated for 15 min then incubated in the fixative for 3 h at room temperature). Fixed tissues were passed through a graded butanol series (50, 70, 85, 95, 100 % butanol), three changes of pure butanol, a 1 : 1 mixture of butanol paraffin oil, and a minimum of six wax changes before embedding in Paraplast Plus. Sections 8 µm thick were cut using a microtome, affixed in poly-l-lysine-coated slides and dried at 42 °C overnight. Digoxigenin-labelled sense and antisense probes were synthesized from a 1·1 kb LjSTM-like cDNA using SP6 and T7 polymerases according to the manufacturer's protocol (Roche). An antisense L. japonicus actin probe was used as a positive control to determine mRNA abundance in the tissues. Microscope slide sections were rehydrated in ethanol series and taken through pre-hybridization treatments according to a modified protocol (Drews et al., 1991; Jackson, 1991). The slides were hybridized overnight at 45 °C in a hybridization solution of 50 % formamide, 300 mm NaCl, 10 mm Tris (pH 7·5), 1 mm EDTA, 5 % dextran sulfate, 1 % blocking reagent and 150 µg ml−1 tRNA. The slides were washed and prepared for immunodetection following the manufacturer's protocol (Roche). Finally, the slides were washed in saline solution and mounted with an aqua-based mounting medium (Shandon Immu-mount).
Identification and characterisation of putative Lotus branching mutants
Lotus japonicus seeds (SS3343) were treated with ethylmethyl sulfonate (EMS) solution at concentrations of 0·4 % (16 h), 0·8 % (3 h), 1·0 % (3 h) or 0·4 % (3 h) (2976 seeds per treatment), sown in soil trays and grown in glasshouse conditions. Ten seed pods were collected from each of the 5928 surviving plants, and the M2 seeds were extracted and packaged. Twelve M2 seeds from each plant were sown into seedling trays and grown in glasshouse conditions. Screening of the M2 progeny plants commenced about 2 weeks after seedling emergence, and putative mutants were transplanted into individual planter bags when the seedlings were well established. M3 seeds of the putative mutant plants were collected, sown and these M3 progeny plants were used for the initial morphological analysis.
Initial genetic analysis was done for a ‘super-accessory’ (sac) shoot branching mutant plants by cross-pollinating the mutant plants with the wild type and determining the wild type : mutant segregation ratio in the F2 population.
RESULTS
Shoot branching pattern in Lotus japonicus
There are two distinct patterns of shoot branching in L. japonicus, set apart by the position on the main shoot from which the axillary shoots arise, basal or aerial shoot branching. Basal branching refers to the branching pattern observed in the cotyledonary region, and aerial branching to the development of branches above the cotyledon.
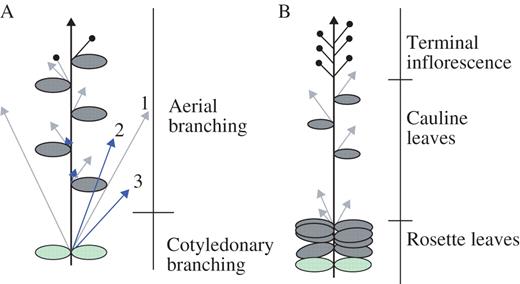
Figure 1 provides a schematic comparison of the branching architectures of wild-type Lotus (Fig. 1A) and wild-type Arabidopsis (Fig. 1B) plants. In wild-type Arabidopsis, axillary shoots usually develop in the axils of rosette and cauline leaves but not in the cotyledons (Grbic and Bleecker, 2000). In Lotus, axillary shoots develop in the axils of all leaves and the cotyledons, throughout the vegetative and reproductive development of the plant. Additional axillary shoots, referred to as accessory shoots, also develop alongside the primary axillary shoots.
Diagram showing wild-type Lotus (A) and wild-type Arabidopsis (B) branching architecture. The primary shoot is indicated by a black arrow. The primary axillary buds/shoots are indicated by grey arrows. Accessory buds/shoots are indicated by blue arrows. Floral meristems are indicated by closed circles. Cotyledons are indicated by green ovals, and leaves by grey ovals. Numbers indicate the order of emergence of axillary and accessory buds.
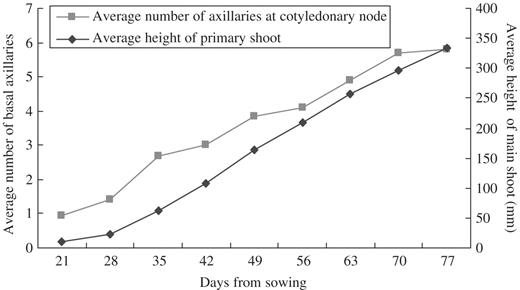
In Lotus, the first pair of axillary buds, referred to as primary axillary buds, is morphologically visible in the cotyledon axils early in the vegetative development of the plant (Fig. 2A). These buds develop into lateral shoots as soon as they are initiated, without being subject to any apparent inhibition exerted by the primary SAMs (Fig. 2B). Additional axillary buds, referred to as accessory buds, continue to develop in the axils of cotyledons, repeating largely the development pattern of the primary shoot, resulting in numerous lateral shoots (Fig. 2C). A new accessory bud forms on the stem side of the cotyledon axil at the basal region of the next older bud (Fig. 2D) and further accessory buds continue to form in this cotyledonary node axil region, long after the cotyledons have abscised. Cotyledonary accessory buds form throughout the growth of the plant, starting very early when the seedlings are barely 6 cm in height (Fig. 3). New accessory buds appear at regular time intervals (at approx. 10 d intervals, Fig. 3).
Shoot branching patterns in Lotus japonicus. (A) A 2-week-old Lotus seedling. (B) A 5-week-old plant. (C) A mature Lotus shoot. (D) Cotyledonary node of a 5-week-old plant; the cotyledon has been abscised. (E) A 3-week-old seedling. (F–H) A mature Lotus plant showing (F) node 1, (G) node 5, (H) node 6. ps, primary shoot; axb, primary axillary bud; axs, primary axillary shoot; acb, accessory bud; acs, accessory shoot; cn, cotyledonary node; cot, cotyledon; fb, flower branch.
Number of axillary buds/shoots at the cotyledonary node and average height of Lotus throughout 77 d growth duration.
Above the cotyledons, primary axillary buds are morphologically visible in the axils of leaves early in the vegetative development of the plant (Fig. 2E). Accessory buds also form in the aerial axils of Lotus (Fig. 2F and G), but usually only a single axillary bud in a leaf axil develops into a lateral shoot, the other accessory buds remaining dormant. However, if accessory buds formed at aerial nodes do develop into lateral shoots, this occurs mostly in the axils closest to the cotyledon (Fig. 2F).
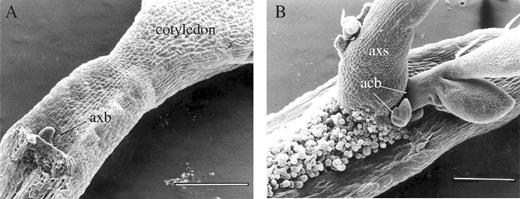
The pattern of shoot branch development in the cotyledonary node region appears to be reiterated in culture-grown detached cotyledons (Fig. 4). Cotyledons were detached from 1-week-old culture-grown seedlings and allowed to grow in a tissue culture medium to observe the proliferative branching pattern. At the time that the cotyledon was detached, a primary axillary bud had already initiated at the cotyledon axil and came off with the detached cotyledon stalk (Fig. 4A). Within 2 months of culture, accessory buds continued to be initiated at the basal region of the primary axillary bud (Fig. 4B). All these buds developed into shoots in a pattern similar to that of the cotyledonary buds in an intact seedling. No other buds formed in any other part of the detached cotyledon.
SEM images showing (A) a newly detached cotyledon and (B) a detached cotyledon after growth in culture medium for 2 months. New accessory buds are indicated by arrows. axb, primary axillary bud; axs, primary axillary shoot; acb, accessory bud. Scale bars in (A) and (B) are 400 μm.
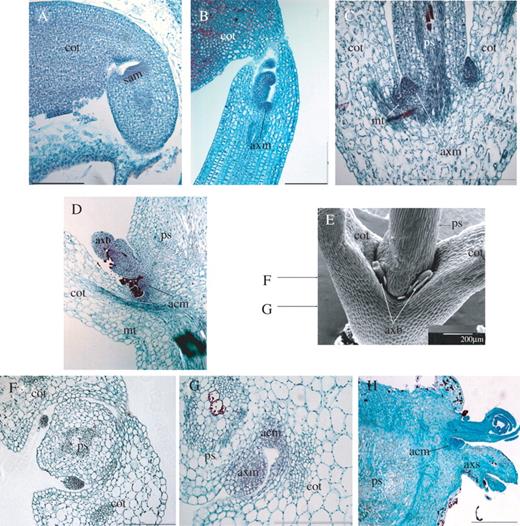
Axillary meristems originate from clusters of densely staining cells
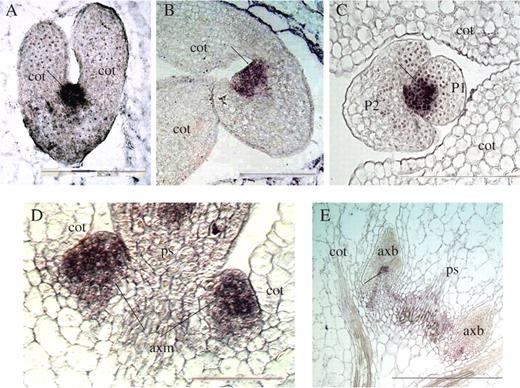
In a mature embryo of Lotus, the SAM appears as a mound of smaller, less vacuolated, densely stained cells (Fig. 5A). Significantly, a portion of this group of densely stained cells extends beyond the apical dome to include a flanking zone that remains in direct continuity with the SAM. Upon seed germination, an axillary meristem is anatomically visible as a group of densely staining cells in the axil of a cotyledon (Fig. 5B, C). Furthermore, groups of densely staining cells are positioned where additional axillary meristems subsequently initiated (Fig. 5C, D). Accessory meristems develop between the axillary bud and the cotyledon (Fig. 5D, G). In a more mature plant, when the cotyledons have abscised, additional accessory buds develop in between the basal region of the next two older accessory buds (Fig. 5H).
Longitudinal sections of (A) mature embryo, (B) newly imbibed seed, (C) 4-day-old seedling and (D) 10-day-old seedling, showing densely staining meristematic tissues. (E) SEM of a 7-day-old seedling Arrows indicate the relative positions of transverse sections shown in (F) and (G). (H) A transverse section through the cotyledonary node of a more mature seedling. sam, shoot apical meristem; cot, cotyledon; axm, axillary meristem; axb, primary axillary bud; axs, primary axillary shoot; acm, accessory meristem; acb, accessory bud; ps, primary shoot, mt, meristematic tissues. Scale bars in A–G, 200 μm; H, 500 μm.
Lotus japonicus SHOOT MERISTEMLESS-like gene transcript accumulation marks meristematic cells
An LjSTM-like cDNA was amplified by RT–PCR using primers homologous to the 5′ and 3′ ends of the open reading frame (ORF) of an M. truncatula knotted-like cDNA sequence. The LjSTM-like translated peptide has 51·5 % identity with STM, and 71 % identity with Medicago knotted-like translated peptide (Fig. 6). In situ hybridization demonstrated that LjSTM-like mRNA accumulates specifically in the SAM of Lotus. In longitudinal sections of Lotus at different stages of development (Fig. 7A, B, D, E) and a transverse section of a young shoot apex (Fig. 7C), LjSTM-like transcripts localize in the SAM. This characteristic expression of the Lotus STM mRNA is similar to the expression pattern observed with STM in Arabidopsis (Long and Barton, 2000). The high sequence similarity of the putative LjSTM-like gene to Arabidopsis STM and the similar meristem-specific patterns of expression in Arabidopsis and Lotus is the basis for using the expression pattern of LjSTM-like gene transcripts to describe the pattern of axillary meristem development in Lotus. In Fig. 7B, LjSTM-like mRNA accumulates in a broad area in the SAM of a mature embryo. A group of cells showing LjSTM-like mRNA accumulation becomes separate from the SAM and is located in the cotyledon axil. LjSTM-like transcripts accumulated in the subepidermal cellular layers in the axil of the cotyledon as shown by a longitudinal section of a 2-day-old seedling (Fig. 8B, C). The accumulation of LjSTM-like transcripts in these groups of cells in the cotyledon axils is associated with initiation sites of primary axillary meristems (Fig. 8C). As the axillary meristems developed into buds, LjSTM-like transcript accumulation persisted in the cotyledon axils, at the basal region of the primary axillary bud, coinciding with the positions where accessory meristems subsequently arise (Fig. 8D). As leaf primordia were formed in these axillary buds, LjSTM-like transcript expression reiterated the pattern of expression in the primary SAM (Fig. 8D). No transcript accumulation was observed in the newly formed leaves of the axillary bud (Fig. 8E). As a negative control, tissues hybridized with the sense probe showed no staining (Fig. 8F). In a transverse section of a more mature Lotus seedling, LjSTM-like transcripts accumulate in cells adjacent to the basal region of axillary or accessory buds (Fig. 8G and H).
(A) Comparison of the deduced amino acid sequence for the LjSTM-like gene (Lj S53343) with that of STM (Long et al., 1996) and the Medicago knotted 1-like gene (Koltai et al., 2001). Vector NTI was used to make the alignment and the deduced distance tree. Identical residues are darkly shaded. Similar residues are lighly shaded. Dashed lines indicate gaps introduced by the programme to attain maximum alignment. (B) Distance tree of different STM cDNAs.
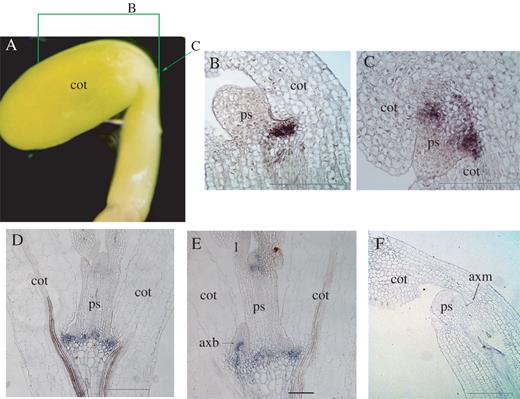
LjSTM-like mRNA (indicated by dark staining) accumulates in shoot apical meristems of Lotus japonicus. Longitudinal sections of (A) late heart/early torpedo, and (B) bent stage embryos showing LjSTM-like transcript accumulation in the presumptive shoot apical meristems (arrows). (C) A transverse section of a young Lotus seedling showing LjSTM-like transcript accumulation in the SAM (dark stained group of cells indicated by an arrow). P1 and P2 leaf primordia curve around the SAM. Longitudinal sections of (D) a 4-day-old seedling showing transcript accumulation in the axillary meristems. (E) In a 7-day-old seedling, transcript accumulation is restricted to the shoot apical meristem of an axillary bud. cot, cotyledon; ps, primary shoot; axm, axillary meristem; axb, axillary bud; P1 and P2, leaf primordia where P2 is older than P1. Scale bars in (A–C) 200 μm; (D) 100 μm; and (E) 200 μm.
Localization of LjSTM-like mRNA (indicated by dark staining) in Lotus. (A) A 2-day-old seedling. A plane and a green line indicate the positions of a sagittal longitudinal section as shown in (B) and a transverse section as shown in (C), respectively. In situ hybridization shows that LjSTM-like transcripts accumulate in the cotyledon axils of a 7-day-old seedling as shown in adjacent median longitudinal sections (D) and (E). As the bud develops, LjSTM-like transcript accumulation is restricted to the shoot apical meristem of the new axillary bud as shown in (E). (F) Longitudinal section of a 2-day-old seedling hybridized with a sense probe. sam, shoot apical meristem; ps, primary shoot; axm, primary axillary meristem; axb, primary axillary bud; axs, primary axillary shoot; acb, accessory bud; acs, accessory shoot; cot, cotyledon; mt, meristematic tissues). Scale bars in (B–D) are 200 μm and that in (E) is 100 μm.
Lotus japonicus mutants with altered shoot branching patterns
In order to identify genes that control shoot branching in Lotus, an EMS-mutagenized Lotus population was screened for mutants altered in accessory axillary branch formation. A mutant plant with increased branching from axillary/accessory buds was isolated from a population of approx. 70 000 M2Lotus seedlings, named sac, for ‘super accessory branches’. Segregation inheritance analysis shows that sac is a recessive mutation (data not shown). Figure 9 shows that compared with the wild type (Fig. 9A), sac (Fig. 9B) had a highly branched phenotype. sac had formed more cotyledonary accessory shoot branches (Fig. 9D) and more aerial accessory buds (Fig. 9G, H) than the wild type. At the end of a 3 month growth period, there were more cotyledonary shoot branches in sac (20) than in the wild type (11) (Table 1). However, the axillary shoots of sac were much shorter in length compared with the wild type (Table 1).
Phenotype of the Lotus super-accessory branching (sac) mutant. Mature (A) wild-type, (B) sac, 77 d after planting (DAP. Cotyledonary nodes of (C) wild type and (D) sac, 2 months after planting. (E–H) Node 1 of Lotus plants, wild type at (E) 35 DAP and (F) 77 DAP, sac at (G) 35 DAP and (H) 77 DAP. Numerous accessory shoots develop in node 1 of sac1 (G) at 77 DAP, and accessory shoots are not distinct from the primary axillary shoot, hence these buds/shoots were not labelled. ps, primary shoot; axs, primary axillary shoot; acb, accessory bud; numbers 1–6 refer to accessory buds according to the order of emergence.
Comparisons of lateral shoot branch development in wild-type and sac Lotus branching mutant, 77 d after planting
| . | Wild type . | sac . |
|---|---|---|
| Mean number of lateral buds+shoot branches | ||
| Cotyledon | 11·1 ± 0·3 | 20·1 ± 0·9 |
| Node 1 | 4·3 ± 0·2 | 8·0 ± 0·4 |
| Node 2 | 3·5 ± 0·1 | 6·3 ± 0·2 |
| Node 3 | 3·5 ± 0·2 | 6·0 ± 0·3 |
| Mean number of nodes/primary shoot | 21·4 ± 0·2 | 17·9 ± 0·4 |
| Mean shoot length of primary axillary shoot | ||
| Cotyledon | 376·7 ± 11·6 | 180·0 ± 12·7 |
| Node 1 | 282·0 ± 30·6 | 140·5 ± 21·4 |
| Node 2 | 222·5 ± 97·0 | 131·7 ± 14·3 |
| Node 3 | 278·3 ± 14·4 | 150·2 ± 12·6 |
| Node 4 | 270·0 ± 15·3 | 117·5 ± 12·03 |
| . | Wild type . | sac . |
|---|---|---|
| Mean number of lateral buds+shoot branches | ||
| Cotyledon | 11·1 ± 0·3 | 20·1 ± 0·9 |
| Node 1 | 4·3 ± 0·2 | 8·0 ± 0·4 |
| Node 2 | 3·5 ± 0·1 | 6·3 ± 0·2 |
| Node 3 | 3·5 ± 0·2 | 6·0 ± 0·3 |
| Mean number of nodes/primary shoot | 21·4 ± 0·2 | 17·9 ± 0·4 |
| Mean shoot length of primary axillary shoot | ||
| Cotyledon | 376·7 ± 11·6 | 180·0 ± 12·7 |
| Node 1 | 282·0 ± 30·6 | 140·5 ± 21·4 |
| Node 2 | 222·5 ± 97·0 | 131·7 ± 14·3 |
| Node 3 | 278·3 ± 14·4 | 150·2 ± 12·6 |
| Node 4 | 270·0 ± 15·3 | 117·5 ± 12·03 |
The mean number of lateral buds+shoot branches (± s.e.) was calculated from 12 sample plants. The mean shoot length (mm) was measured from four sample plants.
Comparisons of lateral shoot branch development in wild-type and sac Lotus branching mutant, 77 d after planting
| . | Wild type . | sac . |
|---|---|---|
| Mean number of lateral buds+shoot branches | ||
| Cotyledon | 11·1 ± 0·3 | 20·1 ± 0·9 |
| Node 1 | 4·3 ± 0·2 | 8·0 ± 0·4 |
| Node 2 | 3·5 ± 0·1 | 6·3 ± 0·2 |
| Node 3 | 3·5 ± 0·2 | 6·0 ± 0·3 |
| Mean number of nodes/primary shoot | 21·4 ± 0·2 | 17·9 ± 0·4 |
| Mean shoot length of primary axillary shoot | ||
| Cotyledon | 376·7 ± 11·6 | 180·0 ± 12·7 |
| Node 1 | 282·0 ± 30·6 | 140·5 ± 21·4 |
| Node 2 | 222·5 ± 97·0 | 131·7 ± 14·3 |
| Node 3 | 278·3 ± 14·4 | 150·2 ± 12·6 |
| Node 4 | 270·0 ± 15·3 | 117·5 ± 12·03 |
| . | Wild type . | sac . |
|---|---|---|
| Mean number of lateral buds+shoot branches | ||
| Cotyledon | 11·1 ± 0·3 | 20·1 ± 0·9 |
| Node 1 | 4·3 ± 0·2 | 8·0 ± 0·4 |
| Node 2 | 3·5 ± 0·1 | 6·3 ± 0·2 |
| Node 3 | 3·5 ± 0·2 | 6·0 ± 0·3 |
| Mean number of nodes/primary shoot | 21·4 ± 0·2 | 17·9 ± 0·4 |
| Mean shoot length of primary axillary shoot | ||
| Cotyledon | 376·7 ± 11·6 | 180·0 ± 12·7 |
| Node 1 | 282·0 ± 30·6 | 140·5 ± 21·4 |
| Node 2 | 222·5 ± 97·0 | 131·7 ± 14·3 |
| Node 3 | 278·3 ± 14·4 | 150·2 ± 12·6 |
| Node 4 | 270·0 ± 15·3 | 117·5 ± 12·03 |
The mean number of lateral buds+shoot branches (± s.e.) was calculated from 12 sample plants. The mean shoot length (mm) was measured from four sample plants.
A different pattern of shoot branching was observed above the cotyledons (Fig. 9E–H). Figure 9E shows a 35-day-old wild-type plant with a single axillary shoot developing at node 1. An accessory bud was initiated in the same axil, but its growth appears to be inhibited (Fig. 9E). In contrast, aerial accessory shoots developed in sac (Fig. 9G, H). At 35 d after sowing, two lateral shoots are developing in sac (Fig. 9G). At 77 d after sowing, numerous accessory shoots have developed in the same leaf axil (Fig. 9H). Because of the formation of more but shorter axillary shoots in both the cotyledonary node and the aerial nodes, sac appears very bushy and dwarf compared with the wild type (Fig. 9A, B).
DISCUSSION
The shoot branching pattern in Lotus differs in the cotyledonary node region from the rest of the plant. In the cotyledonary node region, additional axillary shoot branches (accessory shoot branches) continually form throughout the development of the plant, whereas in the aerial nodes (all nodes above the cotyledons), any additional accessory buds that form usually remain dormant. This proliferative shoot branching pattern in the cotyledonary node makes shoot branching in Lotus very different from that of other plants such as Arabidopsis, where axillary shoots rarely develop in the cotyledon axils (Grbic and Bleecker, 2000) and where axillary shoots usually develop in the axils of rosette leaves only after the primary SAM undergoes the transition to flowering (Grbic and Bleecker, 1996). In another model legume, pea, axillary buds develop at most nodes including the cotyledon axils, but these often remain dormant (Gould and Cutter, 1987; Beveridge et al., 2003). Development of these dormant buds into lateral shoot branches occurs only after environmental and/or developmental constraints are overcome (Stafstrom and Sarup, 2000; Rameau et al., 2002). In petunia, axillary meristems are produced early in the cotyledon axils of the wild type but do not develop into branches (Snowden et al., 2005).
Morphological and histological analyses show that in the cotyledonary axils of an intact Lotus plant, accessory buds appear to form from cells between the basal region of the next two older buds in a pattern very much like the one described in pea (Stafstrom and Sarup, 2000).
There are two hypotheses concerning the developmental origin of axillary meristems. One view maintains that axillary meristems initiate ‘de novo’ in leaf axils (Snow and Snow, 1942; Sharman, 1945) and the opposite view maintains that they are derived from groups of meristem cells detached from the SAM that never lose their meristematic identity (Garrison, 1955; Remphrey and Steeves, 1984). Studies have been done to resolve this issue by using molecular markers. For example, since STM is expressed in all kinds of SAMs, STM expression has been used as a marker of axillary meristem development in Arabidopsis (Grbic and Bleecker, 2000; Long and Barton, 2000). The analysis of Grbic and Bleecker (2000) of axillary meristem ontogeny supports the detached meristem theory. The present observation shows that in Lotus, dense cytoplasmic staining and LjSTM-like transcript accumulation also characterize meristematic cells in the SAM and in groups of cells flanking the SAM, during late embryo development. These groups of meristematic cells are initially restricted to a subepidermal location, consistent with previous observations that STM mRNA transcript accumulates in the L2/L3 cellular layers and is absent from the epidermal cell layer early in the development of axillary meristems in Arabidopsis (Furner and Pumfrey, 1992; Irish and Sussex, 1992; Grbic and Bleecker, 2000). As the axillary meristems developed into dome-like protrusions in the axils, LjSTM-like mRNA accumulation extended to all cellular layers, and as the axillary meristem development progressed into bud development, LjSTM-like mRNA accumulation becomes restricted to a group of cells comprising the new functional SAM. These observations, and their similarity to findings in Arabidopsis, seem to indicate that basal axillary shoot meristems in Lotus are more likely to be formed from ‘detached meristem’ cells rather than being initiated de novo from differentiated cells. However, further study needs to be done to establish that the densely stained STM-like accumulating cells, associated with sites of axillary meristem initiation, continuously had a meristematic fate.
The ability of the cotyledonary region to form additional axillaries throughout the growth of Lotus plant is reiterated in a cultured detached cotyledon, where accessory buds continually form at the cotyledonary stalk, by the side of an existing primary axillary. This observation reveals that shoot branching determinants are probably present in the cotyledon axils even after excision. Whether these are due to the continued persistence of pre-existing meristematic cells is, however, yet to be determined.
The persistence of groups of meristematic cells showing LjSTM-like mRNA accumulation in the cotyledonary node region throughout the development of the plant, even after the cotyledon has abscised, supports the idea that a subset of the meristematic cells is continually set aside as progenitors of new accessory meristems. However, how these meristematic cells are maintained or formed in the cotyledonary node region is yet to be understood. One possibility is that a Lateral Suppressor-like gene has a role in the maintenance of accessory meristem progenitor cells. It has been suggested that the LS gene in tomato (Schumacher et al., 1999) and the LS orthologue LAS in Arabidopsis (Greb et al., 2003) have a role in maintaining the cells set aside for axillary meristems in a pluripotent state.
Previous studies have shown the importance of a subtending leaf in the development of axillary meristems with the connection between the SAM and the adaxial leaf cells being essential for the initiation of axillary meristems (Snow and Snow, 1942; Talbert et al., 1995; McConnell and Barton, 1998). As initiation of cotyledonary accessory meristems in Lotus proceeds long after the cotyledon has abscised, it would appear that the formation of accessory meristems is not dependent on the subtending cotyledons.
The apparent lack of inhibition of axillary and accessory bud outgrowth in the cotyledonary node of Lotus is significant when one considers that in many plant species, the development of axillary and/or accessory meristems into buds and subsequent growth of these buds into shoot branches is usually inhibited by the primary shoot apical meristem or by an existing larger axillary bud (Stafstrom and Sussex, 1992; Stafstrom et al., 1998; Napoli et al., 1999; Shimizu-Sato and Mori, 2001; Beveridge et al., 2003; Horvath et al., 2003; McSteen and Leyser, 2005), often attributed to apical dominance or the inhibitory influence of auxin (as discussed by Cline, 1991, 1996; Chatfield et al., 2000; McSteen and Leyser, 2005).
There appears to be no inhibition of axillary meristem initiation and development in the cotyledonary node region of Lotus by the primary SAM. All of the cotyledonary axillary buds develop immediately into lateral shoots even in very young seedlings, indicating that there may be a variation in the programme regulating cotyledonary accessory shoot branching in Lotus. The regular, proliferative axillary meristem initiation and immediate development of axillary shoots in L. japonicus makes the plant an attractive experimental model for future study of the regulation of axillary meristem initiation and development.
While results of this study are consistent with the notion that axillary meristems originate from detached SAM cells, there remains the possibility that lateral buds forming when the shoot is cut off may arise from differentiated cells. Further analyses of branching mutants, identification and analysis of branching genes and study of hormonal regulation will be necessary to understand the interplay of factors involved in regulating the diverse developmental fate and activity of axillary meristems in L. japonicus.
The isolation of the shoot branching mutant sac provides a step to pursue molecular genetic studies of shoot branching regulation in Lotus. sac mutant plants are characterized by increased proliferation of accessory shoots in all leaf axils including the cotyledons. The proliferative shoot branching pattern observed in the cotyledonary node region of the wild type appears to be reiterated in the aerial nodes of sac. A similar mutation in Arabidopsis, supershoot (sps) is characterized by massive overproliferation of shoots because of an increase in the number of meristems formed in leaf axils and release of bud arrest (Tantikanjana et al., 2001). Whether SAC acts in the same manner as the SPS gene by modulating cytokinin concentration at the site of meristem initiation is a subject for future consideration. The increased rate of accessory shoot production in the cotyledon and increase in the number of accessory shoots in the aerial nodes in the mutants indicate that the SAC gene may negatively regulate accessory shoot meristem initiation and development in Lotus.
We thank Mike Hay, Rod Thomas and Nick Ellison for comments on the manuscript, and Doug Hopcroft and Raymond Bennett of HortResearch for technical assistance in scanning electron microscopy.